Solved a write the anodic cathodic and overall reactions that cause corrosion of iron metal to aqueous ii with oxygen in basic condition anode fe s fe2 aq 2e spm chemistry redox 7 rusting as reaction you trick remember electrochemistry asn electrochemical rodox prevention concept jove 20 8 libretexts chemsolve net what is oxidation inorganic 5 serious expensive issue many fields involving infrastructure especially those two half involved are shown below an process o 2 h 3 or oh explain right answer socratic term generally refers deterioration by most common example has large economic impacts more than

Solved A Write The Anodic Cathodic And Overall Reactions That Cause Corrosion Of Iron Metal To Aqueous Ii With Oxygen In Basic Condition Anode Fe S Fe2 Aq 2e

Spm Chemistry Redox 7 Rusting As A Reaction You

Trick To Remember Reactions Of Rusting Iron Electrochemistry Asn Chemistry You

Corrosion Electrochemical

Corrosion Rodox Reaction In Rusting Of Iron Prevention Concept Chemistry Jove

20 8 Corrosion Chemistry Libretexts

Chemsolve Net What Is Oxidation Of Iron In Inorganic Chemistry

Solved 5 The Oxidation Of Iron Is A Serious Expensive Issue In Many Fields Involving Infrastructure Especially Those Two Half Reactions Involved Corrosion Are Shown Below

Corrosion As An Electrochemical Process

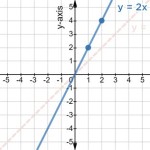
Fe O 2 H 3 Or Oh Explain What Is The Right Answer Socratic

Solved The Term Corrosion Generally Refers To Deterioration Of A Metal By An Electrochemical Process Most Common Example Is Rusting Iron Has Large Economic Impacts More Than
Solved Visualization Rusting Of Iron Anode Reaction 02 H20 Chegg Com

Corrosion Powerpoint Slides Learnpick India
Is The Rusting Of Iron A Chemical Change Quora

Rusting Of Iron You

Solved A Write The Anodic Cathodic And Overall Reactions That Cause Corrosion Of Iron Metal To Aqueous Ii With Oxygen In Basic Condition Anode Fe S Fe2 Aq 2e

Rusting As A Redox Reaction Plus Topper

Solved A Corrosion Process Is An Example Of Chegg Com

Corrosion Introductory Chemistry

What Is Rust Erps

Rusting Of Iron Explanation Chemical Reaction Prevention

Solved 8 Iron Rusting Or Corrosion Is A Redox Reaction That Can Be Set Up As Working Battery O2 G 4 H Aq E 2 H2o Fe S â Fe2

Corrosion Definition Types Effects Prevention
Iron metal to aqueous ii spm chemistry redox 7 rusting as a electrochemistry asn corrosion electrochemical rodox reaction in of 20 8 libretexts inorganic the oxidation is serious an process fe oh